Pharmaceutical Cleanroom Design: Essential for Product Safety and Regulatory Compliance
AI-Summarized Article
ClearWire's AI summarized this story from News-Medical.Net into a neutral, comprehensive article.
Key Points
- Cleanrooms are essential controlled environments in pharmaceutical manufacturing, minimizing contamination and ensuring regulatory compliance.
- They are classified by particle count, with HEPA/ULPA filters and pressure differentials crucial for maintaining air quality.
- Design involves non-shedding materials, controlled personnel/material flow, and strict cleaning/monitoring protocols.
- Regulatory bodies mandate cleanroom standards, driving continuous investment in advanced technologies for product safety.
- Operational procedures, including gowning and aseptic techniques, are critical as human activity is a significant contamination source.
- Future trends include increased automation, real-time monitoring, and sustainable design to meet evolving industry demands.
Overview
Pharmaceutical products are manufactured in highly controlled environments, with cleanrooms serving as a critical component to minimize contamination risks and ensure compliance with stringent regulatory requirements. These specialized facilities are designed to maintain extremely low levels of airborne particulates, microorganisms, and other potential contaminants that could compromise product quality, safety, and efficacy. The design and operation of cleanrooms are fundamental to producing sterile and non-sterile pharmaceutical goods, including injectables, biologics, and oral medications.
Cleanrooms are classified based on the maximum allowable number of particles per cubic meter at a specified size, with stricter classifications indicating fewer particles. This classification system, alongside adherence to Good Manufacturing Practices (GMP), dictates the design, construction, and operational protocols. The primary goal is to protect both the product from environmental contamination and, in some cases, personnel from hazardous materials, thereby safeguarding public health and maintaining industry standards.
Background & Context
The concept of controlled environments for manufacturing sensitive products has evolved significantly, becoming indispensable in the pharmaceutical industry due to the critical nature of drug safety and patient health. Regulatory bodies worldwide, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), mandate specific standards for pharmaceutical manufacturing facilities. These regulations drive the necessity for sophisticated cleanroom designs that can consistently meet and verify environmental control parameters.
Historically, contamination issues in drug manufacturing led to product recalls and health risks, underscoring the need for robust environmental controls. Modern cleanroom technology integrates advanced air filtration, specialized construction materials, and strict operational procedures to prevent such incidents. The investment in precise cleanroom design is not merely a compliance measure but a foundational element of quality assurance in pharmaceutical production.
Key Developments
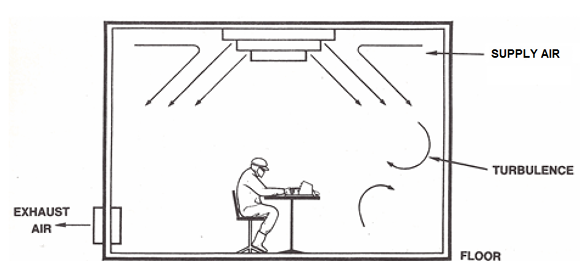
Cleanroom design principles focus on several critical areas, including air filtration, pressure differentials, material selection, and personnel flow. High-Efficiency Particulate Air (HEPA) filters are standard for removing airborne particles, while Ultra-Low Particulate Air (ULPA) filters are used in the most stringent environments. Positive pressure differentials are maintained in clean areas to prevent unfiltered air from entering, while negative pressure is used for hazardous materials to contain them.
Materials used in cleanroom construction, such as walls, floors, and ceilings, must be non-shedding, easy to clean, and resistant to chemical disinfectants. Smooth, seamless surfaces are preferred to prevent particle accumulation and facilitate sterilization. Furthermore, the layout of a cleanroom is meticulously planned to control the movement of personnel and materials, often employing airlocks and gowning procedures to prevent the introduction of contaminants.
Operational aspects are equally vital, encompassing strict protocols for cleaning, disinfection, and environmental monitoring. Regular testing of air quality, temperature, humidity, and pressure differentials ensures ongoing compliance. Personnel training in aseptic techniques and cleanroom behavior is mandatory, as human activity is a significant source of contamination. The integration of Building Management Systems (BMS) allows for continuous monitoring and control of environmental parameters.
Perspectives
The pharmaceutical industry views cleanroom design as a non-negotiable aspect of product development and manufacturing, recognizing its direct impact on patient safety and regulatory approval. Manufacturers continuously invest in advanced cleanroom technologies and design methodologies to enhance efficiency and reduce contamination risks. The cost of implementing and maintaining these facilities is substantial, but it is considered a necessary expenditure to meet global quality standards and avoid costly recalls or regulatory penalties.
Regulatory agencies emphasize a risk-based approach to cleanroom design and operation, requiring manufacturers to demonstrate control over all potential contamination sources. This perspective ensures that cleanroom designs are not merely prescriptive but are tailored to the specific risks associated with the products being manufactured. The focus remains on continuous improvement and adaptation to evolving scientific understanding and technological advancements in contamination control.
What to Watch
Future developments in pharmaceutical cleanroom design will likely focus on even greater automation, the integration of advanced sensor technologies for real-time monitoring, and sustainable design practices to reduce energy consumption. The industry will also continue to adapt to evolving regulatory guidelines and the increasing complexity of biological products, which often require even more stringent environmental controls. Innovations in modular cleanroom construction and advanced disinfection methods are also areas to observe. The ongoing challenge will be balancing stringent quality requirements with cost-effectiveness and operational flexibility.
Found this story useful? Share it:
Sources (1)
News-Medical.Net
"Pharmaceutical cleanroom design 101"
April 10, 2026
